Principle
The pyridaben residues were extracted with Ethyl acetate-Cyclohexane or Acetonitrile, purified by a gel permeation chromatography (GPC) or SPE, determinated by GC-ECD and confirmation by GC-MS. using external standard method.
Reagents and materials
All the reagents used should be analytically pure unless otherwise specified. “Water” is distilled water.
1. Ethyl acetate: HPLC grade.
2. Cyclohexane: HPLC grade.
3. Acetonitrile: HPLC grade
4. Hexane: HPLC grade.
5. Trichloromethane: HPLC grade.
6. Anhydrous sodium sulfate; Ignited at 650 c for
4 h and kept in a tightly closed container.
7. Sodium chloride.
8. Ethyl acetate + Cyclohexane (1+1, V/V).
9. Hexane + Trichloromethane (9+1,V/V).
10. Cyclohexane + Ethyl acetate (6+1, V/V)
11. Pyridaben standard (pyridaben: CAS No. 96489-71-3: molecular Formula C19H25CIN2OS; molecular weight: 364. 93; pure: 99%).
12. Standard stock solution: Accurately weigh a certain amount of pyridaben standard and dis- solve it in a small volume of ethyl acetate. Dilute with Ethyl acetate to make the standard stock solution of 100 mg/L. It should be stored in a refrigerator at 0℃~4℃.
13. Standard working solution: Then dilute the standard stock solution with ethyl acetate to the required concentration as the standard working solution. These solutions should be prepared before use.
14. Carbon-PSA- tube: 1ml, 50mg, active with 3mL acetonitrile + ethyl acetate (10)
15. HLB SPE tubes: 3 mL, 60 mg, or equivalent. Rinse with 2 mL of hexane + trichloromethane
(9), 2 mL of Trichloromethane and 5 mL of distilled water before starting.
16. Film: 0.45 μm.
Apparatus and equipment
1. Gas chromatography: Equipped with ECD
2. Gas chromatography: Equipped with Electron Ionization Mass Spectrometry.
3. Analytical balance: Sense of 0.01 g.
4. Analytical balance: Sense of 0.1 mg
5. Gel permeation chromatograph equipped with isocratic pump and fraction collector.
6. Centrifuge: 4 000 r/min.
7. Rotary vacuum evaporator
8. Anhydrous sodium sulfate column: 7.5 cm x 1.5 cm (i. d.) fuel filled anhydrous sodium sulfate upon 5 cm absorbent cotton. Rotary vacuum evaporator.
9. Vortex mixer.
10. Ultrasonic machine.
11. Homogenizer.
12. 50 mL stoppered plastic centrifuge tube.
Preparation and storage of test sample
Preparation of test sample
1. Cereals and tea
Quarter the sample to ca. 500 g. Grind thoroughly in a high-speed blender. Mix thoroughly and divide into two equal portions as test sample Place in clean containers. seal and label them.
2. Fruits and vegetables
The combined primary sample is reduced to ca. 500, from which shell, seed, peel, stem, root, and coronal has been removed (do not wash by water). Collect the edible parts. and blend and mix thor-oughly in a high-speed blender. Divide into two equal portions. Each portion is placed in a clean container, which is sealed and labeled, as the test sample.
3. Meat and meat products, nuts
Take representative approximately 1 kg of sample. The edible parts are collected. blended and ho- mogenized. Divide into two equal portions. Each portion is placed in a clean container, which is sealed and labeled, as the test sample.
Storage of test samples
The test samples of cereals, tea, nuts, vinegar, honey and honey products should be stored between 0℃~4℃. The test samples of other one should be stored below -18℃. While sampling and sam- ple preparation, precaution must be taken to avoid contamination or any factors that may cause the change of residue content
Procedure
Extraction
1. Vegetables, fruits. cereals
Weight 10 g (accurate to 0. 01 g) of the test sample into a 50 mL stoppered plastic centrifuge tube, then add 20 mL ethyl acetate + cyclohexane extracted with homogenize at 100oor/min for 1 min, then centrifuge at 4000/min for 3 min, transfer the supernatant to another clean tube, and repeat the extraction procedure with 20 mL ethyl acetate + cyclohexane again. The supernatants are passed through a column of anhydrous sodium sulfate to remove the water, collect the effluent into a 150 mL concentrate bottle and condense to nearly dry by a rotary evaporator with a 40 ℃ water bath. Dissolve the residue with 10.0 mL of ethyl acetate cyclohexane, filter through 0. 45 um membrane filter and wait for purification.
2. Walnut. chicken liver. chicken rabbit meat and shrimp
Weight 10 (accurate to 0. 01 g) of the test sample into a 50 mL stoppered plastic centrifuge tube. then add 20 mL acetonitrile, extracted with homogenizer at 10000 r/min for 1 min. then centrifuge at 4 000 r/min for 3 min, transfer the supernatant to another clean tube, and repeat the extraction procedure with 20 mL Acetonitrile again. The supernatants are passed through a column of anhydrous sodium sulfate to remove the water. collect the effluent into a 150 mL concentrate bottle and condense to nearly dry by a rotary evaporator with a 40℃ water bath. Dissolve the residue with 10. 0 mL of ethyl acetate + cyclohexane, filter through 0. 45 um membrane filter and wait for purification.
3. Tea and onion
Weight 2. 5 g (accurate to 0. 01 g) of samples, into a 50 mL stoppered plastic centrifuge tube, add 15 mL distilled water, stand for 1h. then add 20 mL ethyl acetate + cyclohexan, stopper the tubes and vortex for 1 min, then centrifuge at 4000 r/min for 3 min, transfer the supernatant to another clean tube, and repeat the extraction procedure with 20 mL ethyl acetate + cyclohexane again.
The supernatants are passed through a column of anhydrous sodium sulfate to remove the water, collect the effluent into a 150 mL concentrate bottle and condense to nearly dry by a rotary evaporator with a 40℃ water bath. Dissolve the residue with 10.0 mL of ethyl acetate + cyclohexane, filter through 0.45 um membrane filter and wait for purification.
Cleaning-up
GPC Cleaning-up for Vegetables fruits, cereals, walnut, chicken liver. chicken, rabbit meat and shrimp
1. GPC operating condition
a) GPC column: 300 mm 10 mm (i. d.), Bio Beads S-X3 or equivalent:
b) Mobile phase: Cyclohexane-ethyl acetate (1+1):
c) Flow rate: 4. 7 mL/min:
d) Injection volume at sample loop: 5 mL:
e) Time of collecting the eluate: 7.5 min~12. 5 min.
2. GPC Cleaning-up step
Transfer the solution acquired before into the column of GPC with the parameters shown in the first step.
The elution is collected into a clean tube and evaporated to dryness at 35℃ under a stream of nitrogen and redissolved in 1.0 mL ethyl acetate for determination and conformation.
3. SPE Cleaning-up for Vinegar and honey
Weight 2.5 g of samples into a plastic centrifuge tube, add 5 mL of distilled water and vortex for 30 s. Transfer the above solution into SPE column. control the flow at mL/min. Then elute with 2 mL of distilled water. discard the rinse liquid, make vacuum for 2 min. add 4 mL of Hexane + Trichloromethane. collect the eluates into a 10 mL centrifuge tube , then evaporated to dryness at 40 ℃ under a stream of nitrogen ,and dissolved with 0.5 mL ethyl acetate for GC-ECD or GC-MS determina-tion.
4. SPE Cleaning-up for Tea and onion
Transfer the solution acquired before into Carbon- PSA tube, rinse with 1. 5 mL of Cyclohexane + Ethyl acetate. collect the eluates into centrifuge tube, control the flow at 1 mL/min, then evaporated to dryness at 40 ℃ under a stream of nitrogen. and dissolved with 0. 5 mL ethyl acetate for GC-ECD or GC-MS determination.
GC Determination
GC operation conditions
a) Column: DB-1301 30 m 0. 25 mm (i.d.) x 0. 25 um or equivalent;
b) Column temperature: 50℃ (20℃/min) – 200℃ (5℃/min) – 260℃ (10min) .
c) Injection port temperature: 260 C:
d) ECD temperature: 300 C:
e) Carrier gas: nitrogen (purity > 99.999% flow rate: 2 mL/min);
f) Injection mode: Splitless. open the valve after 0.75 min:
g) Injection volume: 1 uL.
GC determination
According to the approximate concentration of pyridaben residues in sample solution, select the standard working solution with similar peak area to that of the sample solution. The standard working solution should be randomly injected in between the injection of sample solution of equal volume.
Under the above GC conditions, the retention time of pyridaben is about 20. 9 min. Standard solution and sample solution were determined according the conditions above, if the sample solution with standard solution with the same retention time peaks then confirmed by GC-MS.
GC-MS confirmation
GC-MS operation conditions
a) Chromatographic column: 30 m x 0. 25 mm (i. d.)
0. 25 um film thickness, and HP-5MS silica capillary column or equivalent;
b) Column temperature: 50 ℃ (hold 1 min) (10℃/min) – 280℃ (10min);
c)Injection port temperature: 250 ℃;
d) Interface temperature: 280 ℃;
e) Electron ionization mode: El;
f) lionization energy: 70 eV:
g) Carrier gas: Helium, purity > 99. 999%, flow rate 1.0 mL/min:
h) Injection mode: Splitless, open the valve after 0.
75 min:
i) Injection volume: 1 uL:
j) Determination mode: SIM:
k) Selected monitoring ions (m/z): 147, 309, 105:
l) Solvent protection delay: 5.0 min.
GC-MS confirmation
According to the operating condition above, analyze the standard solution and sample solution, if there is a peak appeared at the same retention time for both of the sample solution and standard working solution. the GC-MS confirmation test should be conducted. If the retention times of sample chromatogram peaks are consistent with the standard, and after subtracted background noise, the relative intensity ratios of each qualitative ions are also consistent with similar concentration standard. and the similarity degree of their relative abundance ratio in permitted tolerance. we can confirm that there is corresponding analyte in the sample.
Blank test
Blank test will be conducted according to the procedures above without sample addition.
Limit of Determination
The limit of determination of peach, pear, asparagus potatoes, corn buckwheat, honey, vinegar, walnut, rubit meat, chicken meat, shrimp, tea leaf, onion and chicken liver are 0.01 mg/kg.
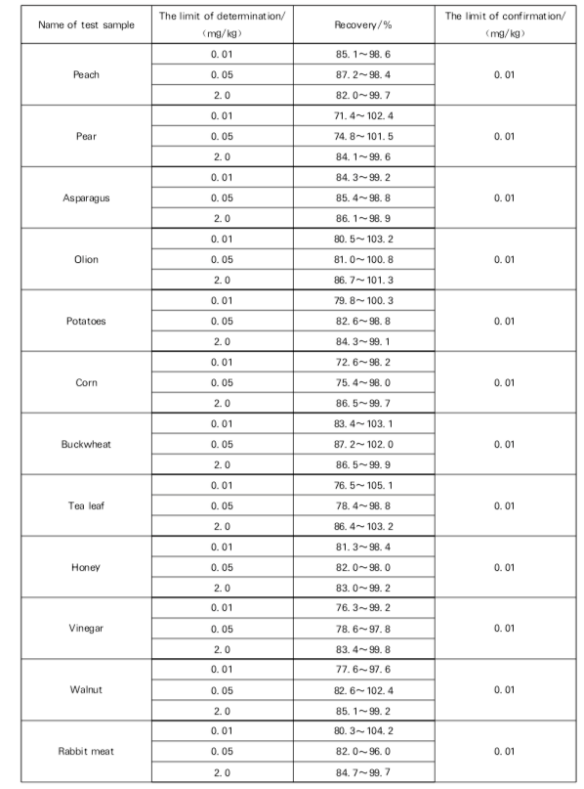
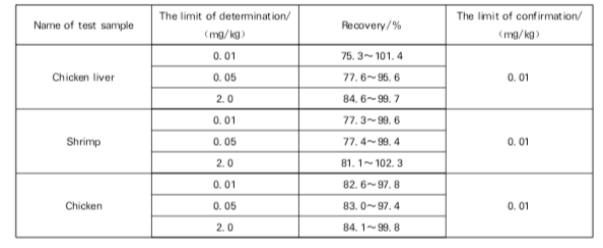
Recovery
The confirmation limit and recovery of this method:
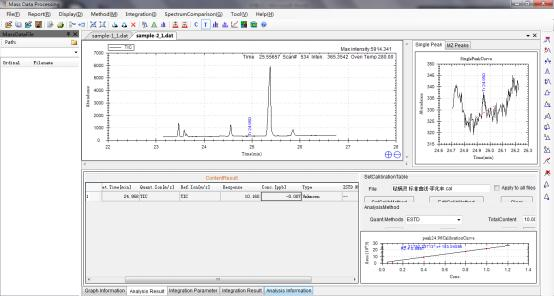
Results by GCMS
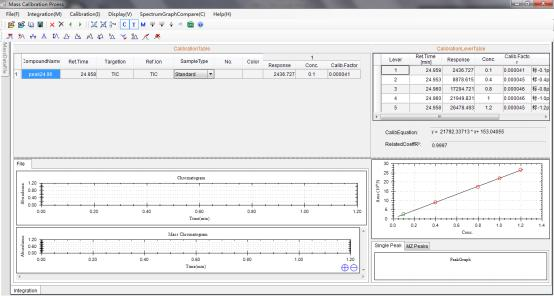
Calibration curve of Pyridaben generated by Standards.
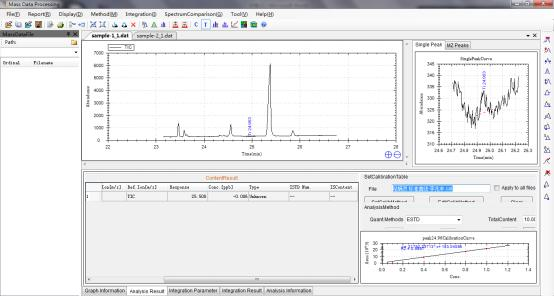
Sample 1:
Sample 2: