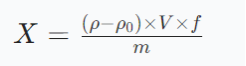Determinação de cálcio em pó-leite – Espectrometria de Absorção Atômica de Flame
- Vista geral dos métodos
Após a amostra ser digerida, a solução de lantano é adicionada como agente de liberação. A amostra é atomizada por chama em um espectrometro de absorção at ômica, e a absorbência é medida a 422,7 nm. Dentro de uma certa gama de concentrações, a absorbância é diretamente proporcional ao conteúdo de cálcio, e a quantificação é realizada em comparação com uma série padrão.
- 2Instrumentos e Reagentes
2.1 Instrumentos e equipamento
2.1.1 Instrumentos de ensaio
Espetrofotometro de Absorção Atômica de Flame (incluir lâmpada de catódio vazio de cálcio)
Compressor de ar (Pressão de descarga estimada: 0,3MPa)
Gas acetileno (pureza do cilindro ≥ 99,99%)
2.1.2 Equipamento de pré-tratamento
Balanço Eletrônico (Sensibilidade: 0,1 mg)
Quartz Crucible(100mL)
Furno elétrico ajustador (Potência estimada: 2000 W)
Muffle Furnace(Temperatura Rated: 1000[UNK])
Plata elétrica quente ajustada (Temperatura estimada: Temperatura do quarto ~ 300[UNK])
Micropipeta(100μL ~ 1000μL, 1000μL ~ 5000μL)
Flasco Volumétrico (100mL, 1000mL)
Tubo colorimétrico(50 mL)
2.2 Reagentes
2.2.1 Reagentes
Ácido nítrico (MOS Grade)
Ácido hidroclórico (Grado Ultra-Puro)
Óxido de lantano (Grado Ultra-Puro)
2.2.2 Reagentes preparados
Solução de ácido nítrico (5 95) Medir 50 ml de ácido nítrico e misturar uniformemente com 950 ml de água. Solução de ácido nítrico (1 1) Medir 50 ml de ácido nítrico e misturar uniformemente com 50 ml de água. Solução de ácido clorídico (1 1) Medir 50 ml de ácido clorídico e misturar uniformemente com 50 ml de água.
- 3Procedimentos Operacionais
3.1 Preparação de amostras
3.1.1 Preparação da Solução de Testes
- Amostra pré -tratamento
Pesa exactamente 2 g da amostra (preciso a 0,0001g) em um crucível de 100 ml de quartz. Coloque em um forno elétrico ajustado e calor suavemente para encaixar até que não seja produzida fumaça. - Transferir para um forno de muffle e cinzas a 550[UNK] por 3-4 horas. Cool e tira. Se a amostra não é completamente cinzasa, adicionar algumas gotas de ácido nítrico, aquecer numa placa quente elétrica ajustada a 150°, para evaporar cuidadosamente, então transferir para o forno de muffle a 550°, para continuar cinzas por 1-2 horas at é a amostra ser cinzas brancas. Cool, tira, adiciona 2 mL de solução de ácido nítrico (1 1), aquecimento em uma placa quente elétrica ajustada a 150[UNK] para dissolver a amostra. Cool, diluir para 50 ml em um tubo colorimétrico com água. Faz um teste em branco de reagente simultaneamente.
- Preparação da solução de amostras de ensaio
Transferir 1 ml da solução de ensaio digerida para um flask volumétrico de 100 ml, adicionar 5 ml de solução de lantano (20g/L), diluir para a marca com solução de ácido nítrico (5 95) e misturar bem. Faz um teste em branco de reagente simultaneamente. A solução de ensaio em branco deve conter os mesmos tipos e quantidades de reagentes que a solução de ensaio, excepto a amostra.
3.1.2 Preparação de soluções padrão
Preparação da série padrão de cálcio
Transferir 0 mL, 0,10 mL, 0,20 mL, 0,30 mL e 0,40 mL da solução padrão de cálcio (1000 μg/mL) em flasks volumétricos de 100 mL, respectivamente. Engadir 5 ml de solução de lantano (20g/L) a cada um, diluir para a marca com solução de ácido nítrico (5 95), e misturar bem. As concentrações desta solução estándar de série de cálcio são 0μg/mL, 1,00μg/mL, 2,00μg/mL, 3,00μg/mL e 4,00μg/mL, respectivamente.
3.2 Ensaios de amostras
- Condições de Teste
Condições de teste de referência para o espectrofotométro de absorção atômica da chama:
Longitude de onda 422,7 nm
Banda espectral de 0,4 nm
Elemento Lampa Corrente 3,0mA
Altura do queimador 10 mm
Taxa de fluxo de gás 1600 ml/min
Pressão do compressor aéreo 0,22 MPa
- Teste de amostra
Introduz as soluções padrão de série de cálcio no atomizador de chama em ordem ascendente de concentração, medir os valores de absorção e trazar uma curva padrão com a concentração de massa de cálcio nas soluções padrão de série como a abscissa e os valores de absorção correspondentes como a ordenada.
Nas mesmas condições experimentais que as para as soluções padrão, introduz a solução em branco e a solução de amostra de teste no atomizador, medir seus respectivos valores de absorção e quantificar em comparação com a série padrão.
3.3 Calculação dos resultados
O conteúdo de cálcio na amostra é calculado usando a seguinte fórmula:
Onde:
X: Conteúdo de cálcio na amostra (mg/kg);
ρ: Concentração em massa de cálcio na solução de amostra de ensaio (μg/ml);
ρ0: Concentração em massa de cálcio na solução em branco (μg/ml);
V: Volume constante da solução de digestão da amostra (ml);
f: Fator de diluição da solução de digestão amostral;
m: Massa de amostra (g).